UDI in Healthcare: What Hospitals Need to Know About Unique Device Identification

Hospitals lose an average of $90,000 each year to expired supplies. During medical device recalls, healthcare organizations spend 16 to 104 hours identifying affected products. These problems share a common root: hospitals can't see what's actually on their shelves.
The solution starts with understanding UDI.
What Is UDI in Healthcare?
UDI stands for Unique Device Identification. It is the FDA-mandated system for identifying and tracking medical devices throughout their lifecycle, from manufacturing to patient use.
Think of UDI as a standardized digital identity for every medical device. Whether it is a surgical implant, a box of exam gloves, or a ventilator, UDI ensures the device can be precisely identified at every point in the healthcare supply chain.
Before UDI, hospitals relied on inconsistent product descriptions, manufacturer catalog numbers, and manual documentation. A single device could have five different identifiers across five different systems. This created confusion, errors, and gaps in traceability.
UDI eliminates that complexity by creating one universal standard.
The U.S. Food and Drug Administration established the system in 2013 as part of the FDA Amendments Act to create a way to rapidly and definitively identify a medical device and its key attributes. Since then, UDI has been adopted globally, with similar systems implemented in the European Union (EUDAMED), China (NMPA), South Korea, and other major healthcare markets.
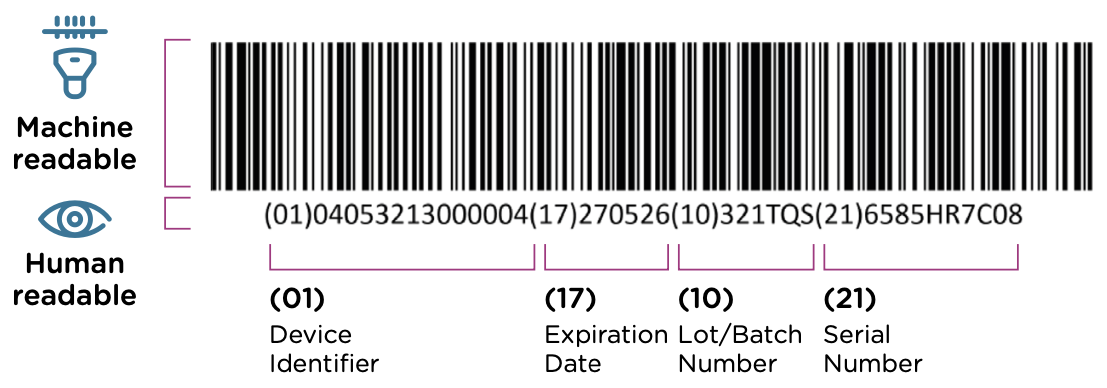
What Does a UDI Look Like? Understanding Device Identifiers and Production Identifiers
Every UDI consists of two parts that work together to create complete device traceability.

Device Identifier (DI): The "What"
The Device Identifier identifies the manufacturer and the specific device model. This portion remains consistent across all units of the same product. Every package of a specific size surgical glove from the same manufacturer shares the same DI.
The DI acts as the reference number that allows hospitals to look up manufacturer information, device specifications, and regulatory details in the FDA's database.
Example DI: 00614141123456 (identifies: Manufacturer X, Model Y, Item Description)
Production Identifier (PI): The "Which One"
The Production Identifier captures what makes each individual unit unique:
- Lot or batch number: Groups devices produced together under the same conditions
- Serial number: A unique identifier for a single device unit (especially critical for implants)
- Expiration date: Prevents use of outdated products
- Manufacturing date: When the device was produced
Example PI: (17)260630(10)ABC12345 (expiration June 30, 2026, lot ABC12345)
Together, the DI tells you what the device is, and the PI tells you which specific unit you are holding.
How UDI Appears on Devices
UDI must be presented in two formats:
- Machine-readable (AIDC): Typically a barcode that can be scanned quickly and accurately. The most common formats are:
- Linear barcodes (GS1-128): Traditional 1D barcodes that work well on larger packaging but become physically long with complex data
- DataMatrix: A 2D square barcode that packs high data density into a small footprint. In an AHRMM survey, 90% of providers supported DataMatrix as the standard UDI barcode format because it fits on small device packaging and can be marked directly on reusable instruments
- QR codes: Primarily used in China's system but uncommon in U.S. healthcare
- Human-readable (HRI): Plain text printed near the barcode so staff can manually read and enter the information if needed.
Where you will find UDI:
- On device packaging (required for all devices)
- On device labels (required for all devices)
- Directly on the device itself (required for reusable devices that are reprocessed between uses)
A hospital receives a shipment of surgical staplers. Each box has a UDI barcode on the label. Scanning that barcode captures: manufacturer (DI), product model (DI), size (DI), lot number (PI), and expiration date (PI). All of this information flows into the hospital's inventory system with a single scan.
Who Issues UDI Codes?
Three FDA-accredited organizations assign UDIs to manufacturers:
- GS1: Dominates with roughly 90% of medical devices using its GTIN-based format. This is the standard you will encounter most frequently.
- HIBCC: Appears on some legacy U.S. manufacturers' products, particularly orthopedic instruments.
- ICCBBA: Manages the ISBT 128 standard exclusively for blood products, tissues, and organs. Hospitals encounter this standard in blood banks and transfusion services.
Is GTIN the Same as UDI? Understanding the Difference
GTIN (Global Trade Item Number) is not the same as UDI, but it is a critical part of it.
GTIN is a product identification number assigned by GS1, the same organization that manages barcodes for retail products worldwide. If you have ever scanned a product at a grocery store, you have encountered a GTIN. In healthcare, GS1 issues approximately 90% of all UDI Device Identifiers, and those Dis are formatted as GTINs.
GTIN serves as the Device Identifier (DI) portion of the UDI for most medical devices. It tells you the manufacturer, the product model, and the package configuration. But GTIN alone does not constitute a complete UDI because a full UDI includes both the Device Identifier (DI) and the Production Identifier (PI). The PI carries the lot number, serial number, expiration date, and manufacturing date that distinguish one specific unit from another. GTIN does not capture any of that production-level detail.
In practice:
- GTIN tells you: “This is a size 7 nitrile surgical glove made by Manufacturer X.”
- Full UDI tells you: “This is a size 7 nitrile surgical glove made by Manufacturer X, from lot ABC12345, expiring June 30, 2025.”

If a hospital only captures the GTIN from a device barcode, it knows what product is on the shelf but cannot determine which specific lot is present, when it expires, or whether it falls under a recall targeting a particular batch. That distinction matters enormously during recall response, expiration management, and implant traceability.
Capturing GTIN is a good start, but capturing the full UDI, both DI and PI, is what unlocks real operational value. GTIN gets you product identification. Full UDI gets you inventory intelligence.
Where UDI Data Lives: The GUDID Database
The FDA maintains the Global Unique Device Identification Database (GUDID), a public catalog that stores DI-level information for all registered medical devices. GUDID now contains over 4 million medical device records.
What is in GUDID:
- Device name and model
- Manufacturer details
- Device classification (Class I, II, or III)
- Product specifications
- Packaging configurations
Who uses GUDID:
- Hospitals: Verify scanned UDI codes against official manufacturer data
- Regulators: Monitor devices on the market and track recalls
- Manufacturers: Maintain accurate product records for compliance
Anyone can search GUDID at AccessGUDID.nlm.nih.gov. Enter a DI, and the database returns the official device record.
Do Hospitals Have to Use UDI? Understanding FDA Requirements
The FDA UDI mandate applies to manufacturers, not hospitals.
What Manufacturers Must Do
Under 21 CFR Part 801 and 21 CFR Part 830, medical device manufacturers are required to:
- Label devices with UDI in both machine-readable and human-readable formats
- Submit device information to GUDID for all devices required to bear a UDI
- Assign new UDIs whenever a device model changes
Compliance dates are based on device risk classification:
- Class III devices (high risk, e.g., pacemakers): September 24, 2014
- Class II devices (moderate risk, e.g., infusion pumps): September 24, 2016
- Implantable, life-supporting, or life-sustaining devices: September 24, 2015
- Class I devices (low risk, e.g., tongue depressors): September 24, 2018-2022 (with some exemptions)
What Hospitals Are NOT Required to Do
The FDA does not mandate that hospitals:
- Scan UDI barcodes at receiving
- Capture UDI data at point of use
- Integrate UDI into EHRs or inventory systems
- Use UDI for internal inventory management
But hospitals that do not capture UDI still face regulatory and operational pressures that make adoption the smart choice:
- Joint Commission requirements: The Joint Commission requires hospitals to track implanted devices in patient records with bidirectional tracing and complete audit trails. UDI is the most efficient way to do this.
- FDA recall compliance: When the FDA issues a recall, hospitals must identify and remove affected devices. Without UDI capture, this process takes days. With UDI, it takes hours. Healthcare providers estimate 16 to 104 hours of labor per recall without UDI, according to a 2021 AHRMM report. With UDI-enabled systems, recall research drops to minutes.
- CMS reimbursement pressure: CMS's TEAMS bundled payment model, set to launch in 2026, will require more precise implant documentation to optimize reimbursement. With 40-60% of hospital reimbursement coming from CMS, even modest payment adjustments tied to UDI documentation could transform adoption economics overnight.
- Internal efficiency goals: CFOs, supply chain directors, and clinical leaders want to reduce waste, prevent expirations, and optimize purchasing. UDI provides the data needed to achieve these goals.
International UDI Requirements
Global UDI requirements are tightening rapidly:
- European Union: EUDAMED UDI/Device registration module becomes mandatory in May 2026
- Australia: TGA implementing phased UDI compliance starting with Class III devices in July 2026
Hospitals are not legally required to use UDI in the U.S. But the hospitals that capture UDI data consistently gain significant operational and financial advantages over those that don’t.
What Can Hospitals Use UDI For? Practical Applications That Drive Value
UDI solves some of the most expensive, time-consuming problems in hospital supply chain management.
1. Reducing Expired Supply Waste
Hospitals lose an average of $90,000 per year to expired supplies, according to GHX inventory count data from 2024, because they do not have real-time visibility into expiration dates. Product expiration alone averages 8-10% of total supply spend. For a hospital spending $100 million on supplies annually, that is $8-10 million wasted on expired products. Staff find expired devices during procedures, pull them from use, and replace them with fresh inventory. But the expired items were already purchased, and now they are waste.
When hospitals scan UDI barcodes during periodic inventory audits, they capture expiration dates automatically. The system can then flag items approaching expiration and trigger alerts to clinical areas: "You have 12 units of Product X expiring in 30 days. Use or relocate these before purchasing more." Organizations with UDI-enabled inventory visibility have reduced expirations to less than 1%, recovering $7-9 million annually in hospitals with $100M supply spend.
2. Improving Inventory Accuracy
Hospital inventory systems often show one thing while the shelf shows another. The system says you have 50 units of a device. The shelf has 35. Where are the other 15? Were they used and not documented? Expired and discarded? Misplaced? Without UDI, no one knows. This inventory inaccuracy forces hospitals to overstock "just in case," tying up millions in working capital.
UDI scanning creates a quality control layer between physical inventory and digital records. When supply chain staff scan devices on the shelf and compare results to what the system shows, discrepancies become visible immediately. A UDI-based baseline inventory also exposes item master problems: products in the system without accurate UDI data, items on the shelf that don't exist in the system at all, and duplicate entries that inflate perceived inventory levels.
3. Speeding Up Recall Response
Medical device recalls require speed and precision. The FDA issues a recall notice. Hospitals must identify affected devices by lot number or serial number, locate them across multiple storage areas, remove them from clinical use, document every action taken, and report results to the FDA.
Without UDI, this process involves manually searching purchasing records, checking multiple inventory systems, physically inspecting storage areas, and coordinating with clinical staff to check preference cards and procedure carts. Average time: 16 to 104 hours, according to a 2021 AHRMM report. Medical device recalls hit a four-year high of 1,059 events in 2024, according to Sedgwick's 2025 Recall Index report, making this an increasingly urgent operational problem.
When hospitals maintain up-to-date UDI snapshots of their inventory and have a UDI-enabled recall partner, such as SxanPro, they can search by lot number or serial number instantly. The system returns: "You have 6 units of the recalled product. They are located in OR 3, OR 7, and the Central Sterile supply room." Staff can remove the devices immediately, quarantine them, notify clinical areas, and document the entire process in a fraction of the time.
4. Strengthening Patient Safety and Implant Traceability
When a patient receives an implantable device (pacemaker, joint replacement, surgical mesh), the hospital must document exactly which device was used. If the device is later recalled or associated with adverse events, the hospital needs to identify which patients received devices from the affected lot. Without UDI, clinicians often rely on manual documentation: handwritten stickers, product labels taped to charts, or manual entry into EHRs. This creates errors, incomplete records, and traceability gaps. An analysis by the International Consortium of Investigative Journalists of FDA adverse event data found that between 2008 and 2017, more than 1.7 million injuries and nearly 83,000 deaths were potentially linked to medical devices, highlighting the importance of accurate device tracking.
Scanning UDI at point of use captures the device identifier, lot number, serial number, and expiration date directly into the patient's electronic health record. This creates a permanent, accurate record of which device was used for which patient.
A patient receives a knee implant. The surgical team scans the UDI barcode before the procedure. The data flows automatically into the patient's EHR. Five years later, the manufacturer issues a recall for devices from that lot. The hospital searches its EHR by lot number and identifies the patient within seconds, enabling timely follow-up.
The ONC requires certified EHR systems to include UDI fields for implantable devices. The infrastructure exists inside Epic, Oracle Health, and Meditech. The fields are built. Hospitals just need to populate them.
5. Recovering Lost Revenue Through Better Charge Capture
The Healthcare Financial Management Association estimates hospitals lose 1-2% of net revenue to charge capture errors. For a hospital generating $200 million in net patient revenue, that translates to $2-4 million in lost reimbursement annually, and the problem compounds across thousands of facilities nationwide. An orthopedic unit performing 20 surgeries daily could forfeit over $1.2 million annually from just four missed implant charges per day. A study published in the Journal of Neurosurgery found that UCSF Medical Center disposed of an average of $968 in unused medical supplies per neurosurgery procedure, totaling $2.9 million in waste from a single department in one year. Poor documentation means these items often are not charged correctly.
Scanning UDI at point of use automatically documents which devices were opened and used, creating an accurate charge record that flows directly to billing systems. But point-of-use scanning only works when the hospital's item master contains accurate UDI data. Without a clean item master, the barcode scan returns no match or an incorrect match, and the documentation benefit disappears.
6. Optimizing Purchasing and Reducing Overstock
Hospitals often purchase more inventory than they need because they do not have accurate data on what is being used, what is sitting unused, and what is expiring. This leads to overstock, cash tied up in inventory, and storage space wasted on slow-moving products. According to industry estimates, hospital supply chain expenses consume approximately 33% of operating budgets, averaging $72 million for a standard hospital.
UDI data reveals true utilization patterns. By tracking which devices are scanned and used versus which devices sit untouched, supply chain teams can identify low-turnover products to reduce par levels, detect duplicate inventory across storage locations, and make data-driven purchasing decisions based on actual consumption.
How Hospitals Implement UDI Scanning: Technology Options
Every medical device in a hospital already carries a UDI barcode. The data is printed right on the label. The challenge has never been the availability of information. It has been capturing that information without requiring massive infrastructure investments, system integrations, or workflow changes that clinical staff resist.
Historically, hospitals approached this problem by adding infrastructure: smart cabinets that detect items automatically, or integrated scanning stations built into receiving and point-of-use workflows. These approaches carry significant costs, ranging from thousands to tens of thousands of dollars per unit, require additional setup and demand facility-wide rollouts to deliver comprehensive coverage. Integrated scanning workflows require custom software connections between hardware, EHR systems, and inventory platforms, creating IT projects that take months or years to implement.
These infrastructure-heavy models also assume hospitals need to track every device movement in real time: scan in, scan out, scan at every transition point. For most hospitals, especially those just beginning their UDI journey, that level of transactional tracking is neither realistic nor necessary.
The more practical question is simpler: what is actually on your shelves right now, and does it match what your system says?
The Problem with Manual Entry
When hospitals attempt to capture UDI data without scanning technology, they default to manual entry. Staff read the human-readable text on device labels and type the information into inventory systems or spreadsheets. This approach introduces errors at a predictable rate, approximately every 3-5 devices keyed in. A single transposed digit in a lot number can make a device invisible to the system. A mistyped expiration date can leave an expired product in circulation or flag a perfectly good device for removal.
Over hundreds or thousands of devices across multiple storage locations, those errors compound into unreliable data that undermines the entire purpose of tracking. Manual entry is also slow, labor-intensive, and unsustainable as a long-term inventory management strategy. Staff already stretched thin in clinical environments cannot be expected to manually transcribe 14-digit GTINs, lot numbers, and date codes without mistakes.
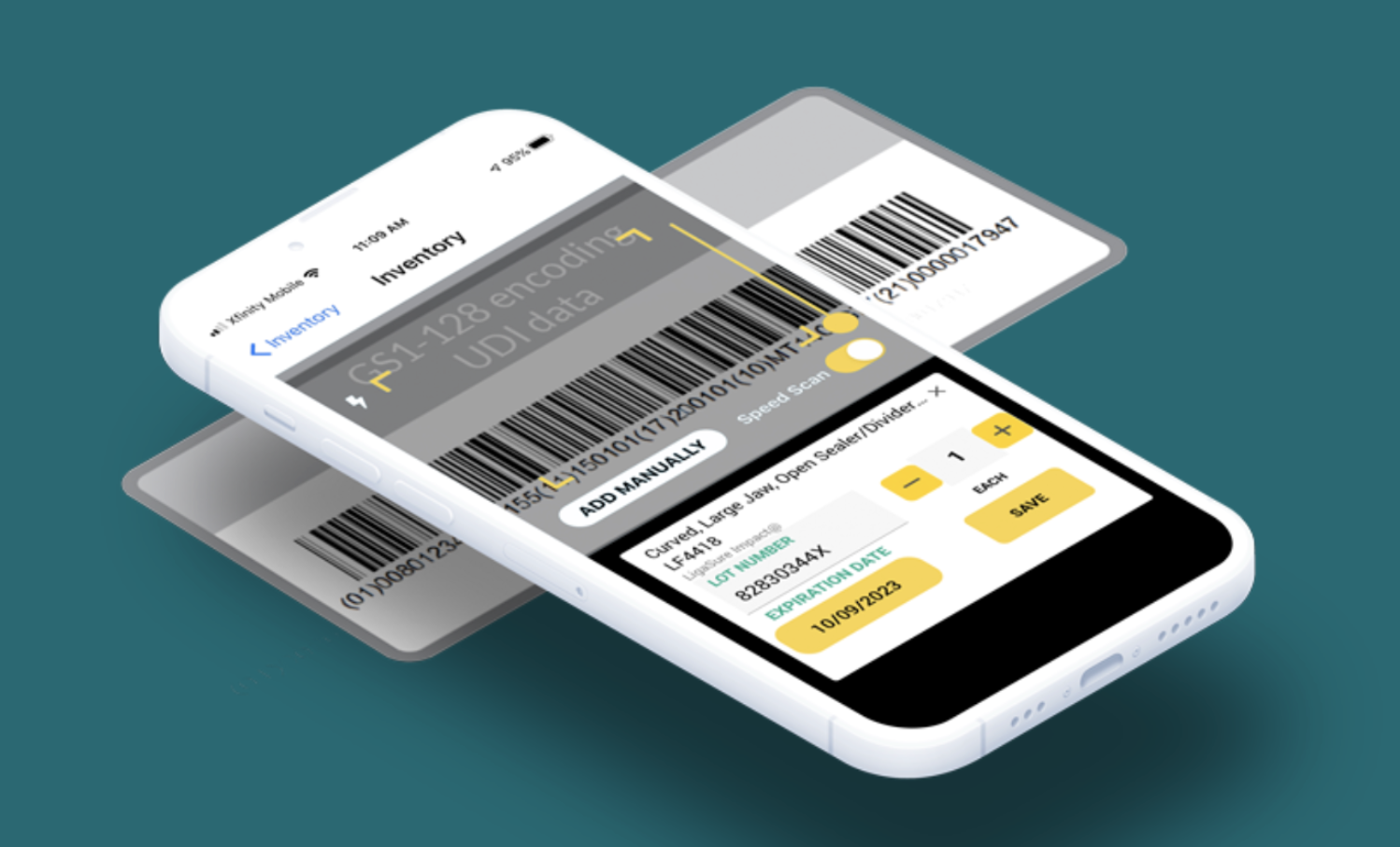
Mobile Snapshot Scanning (SxanPro's Approach)
UDI was designed to make device identification simple: one scan captures manufacturer, product, lot number, expiration date, and serial number. SxanPro's approach takes full advantage of that design.
%20(1).jpeg)
How It Works:
- Baseline inventory without integration: Supply chain staff use mobile devices to scan UDI barcodes directly from devices on the shelf. No integration with the hospital's ERP or inventory management system is required. This baseline scan captures a complete picture of what is physically present and creates the foundation for a clean, UDI-enriched item master.
- Point-in-time snapshot methodology: Instead of requiring real-time transactional scanning at every movement, SxanPro captures periodic snapshots of what is physically present. These can be built into daily workflows without disrupting clinical operations. Each snapshot creates an updated record of inventory that can be compared to system records.
- Gap identification: SxanPro compares physical scan results to the hospital's systems of record and highlights discrepancies: devices that are present but not in the system, devices shown in the system but missing from the shelf, and devices approaching expiration.
- Recall matching: SxanPro maintains a proprietary recall database sourced from the FDA and matches all scanned inventory data against it daily. When a recall is issued, hospitals know immediately whether affected products are on their shelves and exactly where they are located.
- Actionable insights: Hospitals receive dashboards showing inventory health, expiration risk, recall-affected items, and financial recovery opportunities.
Because UDI already exists on every device label, hospitals do not need to add tags, install cabinets, or build integrations to gain inventory visibility. They need a way to read what is already there and turn it into usable data. SxanPro's patented UDI parsing technology does exactly that, extracting the full UDI (both DI and PI) from a scan and translating it into actionable intelligence about what a hospital owns, where it sits, when it expires, and whether it has been recalled. The baseline scan also gives hospitals clean UDI data to enrich their item master, which is the prerequisite for making UDI work at point of use.
Traditional inventory management requires hospitals to integrate new software with existing systems, train staff on complex workflows, and change how receiving, stocking, and usage are documented. This is expensive, time-consuming, and often fails because clinical staff resist workflow changes. SxanPro requires no integration, no workflow disruption, and no changes to how staff currently operate. It adds a quality control layer that makes invisible problems visible.
One SxanPro client recovered $642,938 in cost savings within the first year by identifying at risk inventory, eliminating redundant stock, and optimizing PARs based on UDI scan data.
Why Hospitals Struggle and How to Overcome Resistance
The most comprehensive research on adoption barriers comes from studies across major health systems, confirming that the obstacles are primarily organizational and operational, not technological.
The single largest barrier is the absence of a hospital-side mandate. Without regulatory penalties or reimbursement consequences, UDI implementation competes against every other priority for budget and attention. A 2023 study published in BMJ Open, conducted with eight NESTcc health system network collaborators, found only three had implemented UDI capture for implantable devices, while four had done so in very limited fashion or not at all.
IT infrastructure complexity ranks as the second major barrier. Health systems operate diverse point-of-care IT systems. One study found nine different systems in cath labs and six different systems in ORs across just 10 health systems. Integrating UDI data flow from scanning through EHR to claims requires custom software modifications.
Clinical staff resistance reflects rational concerns about workflow disruption in high-pressure environments. The most effective change management approaches:
- Reframe UDI as a patient safety tool rather than a compliance requirement
- Start with a perioperative or nurse champion who has cross-disciplinary credibility
- Deploy technology that eliminates rather than adds steps
- Pilot in a single controlled department (cardiac cath lab works best, contained environment with high-value implants and dedicated staff)
- Demonstrate measurable wins within 90 days
- Expand methodically based on proven results
AHRMM’s Learning UDI Community has developed Quick Start Guides, case studies, and workflow documentation specifically to shorten the adoption curve for new implementers.
The Future: AI, Real-World Evidence, and What Comes Next
The future of UDI extends far beyond compliance into strategic intelligence. 97% of medical device data currently goes unused, representing a massive untapped resource that AI and machine learning will unlock.
AI-driven demand forecasting using UDI consumption data promises to transform supply chain operations. Johnson & Johnson is already using ML algorithms for demand forecasting, achieving a 15% productivity increase. The healthcare digital twin market, projected to grow from $2.29 billion in 2024 to $77.4 billion by 2034, will enable hospitals to simulate supply chain scenarios and optimize inventory positioning before demand fluctuations occur.
Real-world evidence for medical devices is accelerating. The FDA’s updated December 2025 guidance removed previous requirements for individual patient-level data in submissions, making UDI-linked databases far more practical for post-market surveillance. The NESTcc network now includes 19 collaborators generating real-world evidence, according to a 2024 GAO report, and the BUILD program has demonstrated that coronary stent outcomes can be tracked across multiple health systems using UDI-linked data.
Getting Started: What Hospitals Should Do Next
If your hospital has not started capturing UDI data, this is a practical roadmap:
Step 1: Audit One High-Value Procedural Area. Choose a single high-dollar procedural and perform a UDI-based baseline inventory. Scan every device on the shelf and capture the full UDI, both DI and PI. Measure the discrepancies between what you find and what your system shows. This creates a baseline and demonstrates ROI potential to leadership.
Step 2: Build UDI into Your Item Master. Use the baseline inventory data to clean and enrich your item master with accurate UDI information. When your item master contains reliable UDI data, every downstream process improves: barcodes become scannable at point of use, expiration tracking becomes automated, and recall matching becomes instant. Without a clean item master, point-of-use scanning fails before it starts.
Step 3: Establish a Recall Readiness Protocol. Identify your highest-risk device categories (implants, life-sustaining devices) and partner with a recall management solution that uses UDI. SxanPro maintains a proprietary recall database sourced from the FDA and matches all scanned inventory data against it daily, so hospitals know immediately when a recalled product is on their shelves.
Step 4: Explore Snapshot-Based Solutions. Once your item master is clean and your baseline is established, build UDI scanning into daily workflows through point-in-time snapshots. Tools like SxanPro do not require integration or workflow changes. Periodic snapshot scanning gives you ongoing visibility into inventory health, expiration risk, and recall exposure without the cost and complexity of enterprise-wide system overhauls. One SxanPro client recovered $642,938 in cost savings within the first year by identifying expired inventory, eliminating duplicate stock, and optimizing purchasing based on UDI scan data. Most successful implementations demonstrate ROI within six months.
Additional Resources for Learning More
FDA's UDI System Resources https://www.fda.gov/medical-devices/device-advice-comprehensive-regulatory-assistance/unique-device-identification-system-udi-system
The FDA's official page with comprehensive UDI rule guidance, training materials, and educational resources
AHRMM Learning UDI Community (LUC) https://www.ahrmm.org/resources/learning-udi-community
A coalition of healthcare leaders providing Quick Start Guides, educational webinars, and practical implementation resources
GS1 Healthcare UDI Resources https://www.gs1.org/industries/healthcare/udi Global standards organization offering educational materials and implementation guides for UDI
AccessGUDID Database https://accessgudid.nlm.nih.gov
Search the FDA's public database of UDI device identifiers
Power Supply Interview with Ashlea (SxanPro Founder/CEO) Leveraging the UDI Barcode: The Hidden Truth About Inventory
Learn about practical UDI implementation from industry experts
Sources
AHRMM, "Cost of Recall Management Report" (2021) - 16 to 104 hours per recall
Sedgwick, "2025 U.S. State of the Nation Recall Index Report" - 1,059 recall events in 2024
About SxanPro
SxanPro is a Intuitive patented mobile technology that leverages Unique DeviceIdentification (UDI) to enhance inventory data quality to improve supply chain processes in healthcare settings. By transforming better data into actionable insights, we empower organizations to make informed, data-driven decisions that drive cost recovery and operational efficiency.
.avif)